Dic 15, 2020 | Sin categorizar
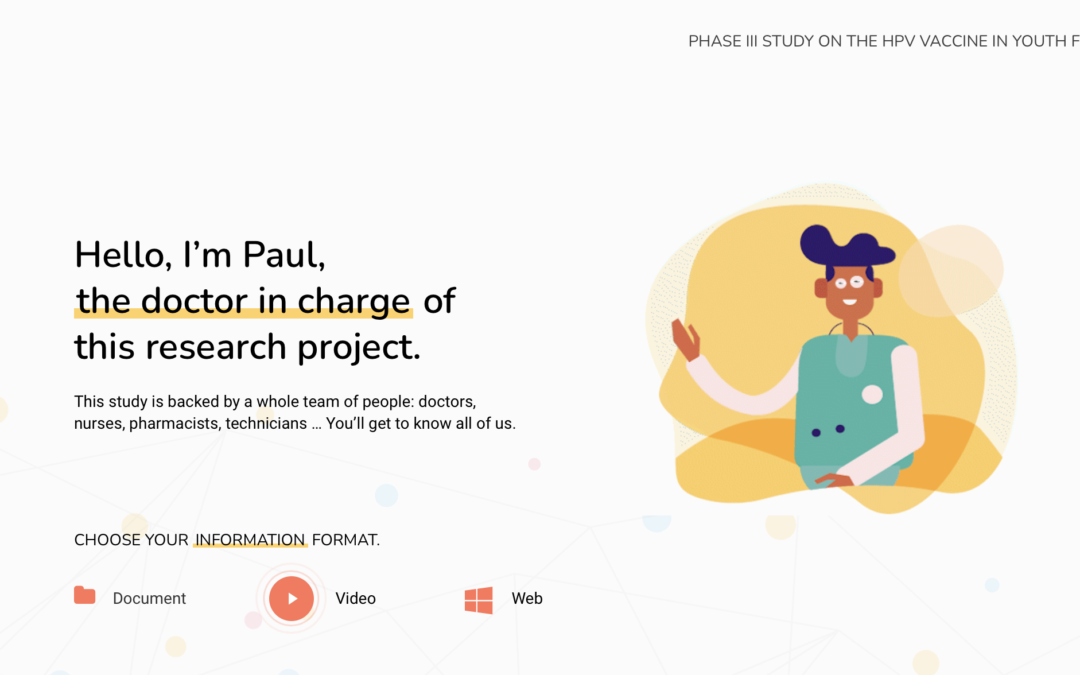
i-CONSENT has elaborated examples of communication materials to be used in clinical trials following the project recommendations. The idea was to elaborate a series of materials to deliver all the...

Dic 10, 2020 | Sin categorizar
The first i-CONSENT informed consent is now circulating. A real Spanish clinical trial has used the project guidelines to elaborate a series of communication materials to provide the information on the study to the participants. The ethics committee has not only...

Dic 1, 2020 | Sin categorizar, Press Release
The FISABIO team sat together with experts in the field and discussed the guidelines’ content using the RAND/UCLA methodology In addition to the validation of the materials elaborated following the i-CONSENT guidelines, it has deemed necessary to...

Nov 26, 2020 | Sin categorizar
Summary of the sessions organized by our partner LUMSA University Following the sessions organised to discuss informed consent for clinical trials in the context of an emergency setting, find a summary of each meetings’ main ideas. Research protocols and...

Nov 16, 2020 | Events, Sin categorizar
i-CONSENT organizes a series of workshops to discuss informed consent for clinical trials in the context of an emergency setting i-CONSENT partner, LUMSA University, has organized a series of internal online workshops to discuss informed consent in the...




