- From WP2 and WP3 to the elaboration of the guidelines
With approximately one year of project left, i-CONSENT is starting to translate the findings into a set of guidelines to improve Informed Consent. The conclusions of i-CONSENT WP1 (Work package 1, A multi-layered approach to informed consent) and WP2 (Work package 2, Innovation in informed consent), will now be shaped by WP3.
The informed consent process is a bidirectional communication process that begins with the first contact with the potential participant and goes through the entire research project until its end. The main scope of the informed consent process is to respect the autonomy of the patient to take decisions about his/her own health, being the information a key aspect that should be easily accessible (provided all relevant information) and understandable; the decision making about participating (or continuing) in a research or not should be free and done without any coercion. This is how i-CONSENT graphically describes the stages of the informed consent process:
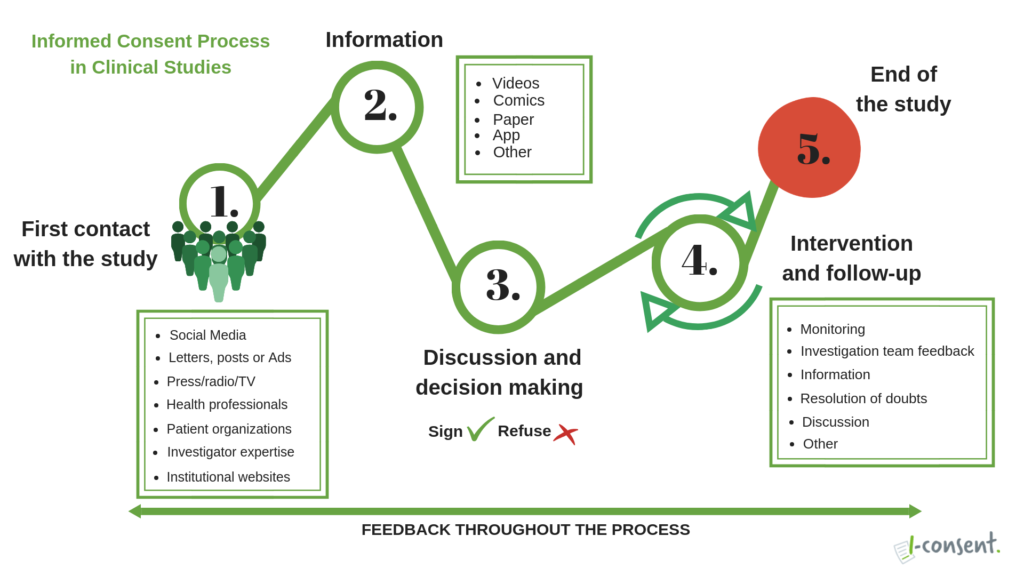
All the stages of the process will be included into the guidelines. The partners’ effort will be devoted to developing recommendations for the final draft of the guidelines, including in particular the use of new technologies (ICT, videos, interactive apps…).
The drafting of the guidelines will take into particular account the core focuses of i-CONSENT:
- Gender issues;
- Ethical issues in translational research;
- ICT based strategies.
WP4 – Communication and dissemination
In the framework of the dissemination strategy of the project, the partners will also prepare scientific publications in order to share with the scientific community the first results of their research within i-CONSENT project.
Check our already published articles:
- ANCEI. La comprensión de los documentos de asentimiento en la investigación con menores: una revisión sistemática. (FISABIO, UCV). Text here.
- ANCEI. Contenidos del asentimiento del menor en investigación médica: Diferencias entre la literatura científica y el requisito legal. (FISABIO, UCV, LUMSA). Text here.
- Medicina y Ética. Ethical issues concerning informed consent in translational/clinical research and Vaccinations bias and informed consent. (UNESCO, GSK). Text here.
- Studia Bioethica. Il Consenso Informato. Informed Consent in Clinical Research: Interdisciplinary and Cross-Cultural Perspectives (UNESCO, LUMSA). Text here.
- ConBioPreVal Book of Abstracts. How to strengthen the recommendations for the informed consent process in health-related studies: The i-CONSENT project methodology. Text here.

More publications will follow along with workshop organisation, campaigns and networking activities. Want to join us? Check our social media and webpage and stay tuned!
